
Keys for Optimizing Orphan Drug Development
Maximizing Clinical Trial Access, Diversity, and Efficiency are Keys to Optimizing Orphan Drug Development February 22, 2022 February is Rare Disease Month—an important time when
Jeeva Trials > News

Maximizing Clinical Trial Access, Diversity, and Efficiency are Keys to Optimizing Orphan Drug Development February 22, 2022 February is Rare Disease Month—an important time when

By Harsha Rajasimha, PhD, Founder and CEO, Jeeva Clinical Trials Inc. www.jeevatrials.com To say that COVID-19 has been a challenge to healthcare systems around the

Clinical trials can save lives and provide new ways to treat disease. The industry estimates that 85% of all clinical trials face delays due to

The loss of his daughter to a rare congenital disorder in the United States and the passing of his younger brother in India from complications

The growing use of decentralized clinical trials has raised serious concerns regarding verification of patient comprehension and proper informed consent. The solution, says Dr. Harsha

Almost everything went online due to the COVID-19 pandemic, from remote collaboration with coworkers to ordering groceries and conducting online classes. But unlike other industries

Diversity is a growing concern in all aspects of modern life, but the ramifications of this disparity in health care can have life threatening consequences.

MANASSAS, VA. (PRWEB) DECEMBER 01, 2021 The ongoing COVID-19 pandemic ranks as perhaps the most serious global health crisis of the past hundred years. Worldwide,

IRBs are seeing a surge in study protocols that include remote recruitment, consent, and participation, raising ethical and regulatory considerations for investigators. “We are seeing

Serious inefficiencies are hampering clinical trials of new drugs and treatments, prolonging the suffering of millions. Today’s platform technologies, says Dr. Harsha Rajasimha of Jeeva

The outbreak of the COVID-19 pandemic seriously disrupted new drug testing, treatments, and in-person healthcare services; access to clinical trial sites was reduced by some

Leaders from the trial tech provider discuss how human-focused technology on eClinical platforms can tackle inefficiencies associated with traditional studies. According to Harsha Rajasimha, CEO

Serious inefficiencies are hampering clinical trials of new drugs and treatments, prolonging the suffering of millions. Today’s platform technologies, says Dr. Harsha Rajasimha of Jeeva

Did you know that 80% of participants of randomized, controlled clinical trials are Caucasian males or that less than 10% of those trials included children?

The clinical trial industry must proactively address a record of “historical errors and misbehavior” in its treatment of minority populations if it wants to rebuild
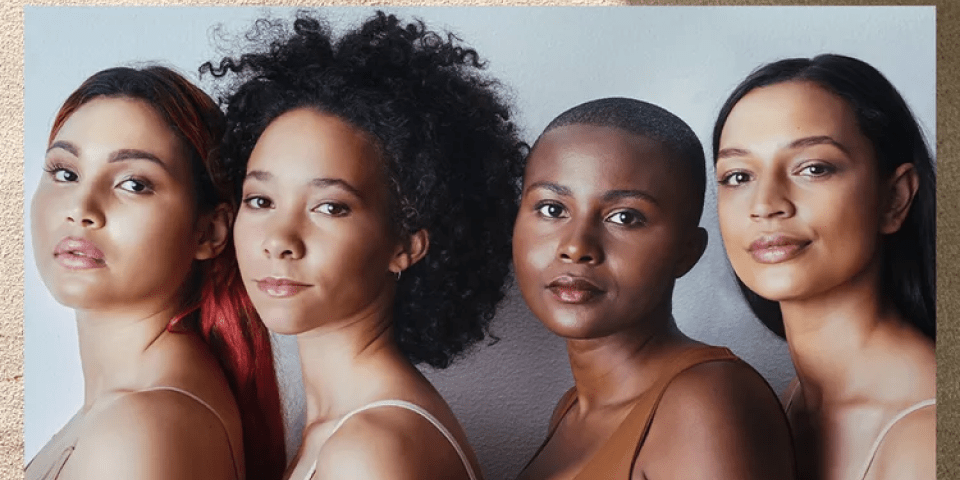
Before medications and treatments can be deemed safe and effective for the masses, they are rigorously studied in a process called clinical trials. During clinical

Clinical trials of new drugs and treatments have a high failure rate. Better planning and technology-aided communication, says Dr. Harsha Rajasimha of Jeeva Trials, can

LUZERNE COUNTY, Pa. — Right now, there are clinical trials for COVID-19 treatments, as well as treatments for all kinds of other medical issues. The

“It’s not reasonable, ethical, or fair to expect patients to go to a physical site when it’s not necessary” for the conduct of a clinical

Over the last two years, testing of new drugs, medical devices, and vaccines has increasingly been done outside large medical centers. To benefit both sponsors
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
Dr. Sweta Sneha is the Founder and Executive Director of Healthcare Management & Informatics and Professor of Information Systems & Security at Kennesaw State University. A globally recognized expert with a passion towards equity and diversity, Sneha is a strategic leader with two decades of experience in technology and informatics. She is a recipient of Distinguished Service Award, KSU Collaboration Award, NCWIT RISE-IT Award, and WIT Honoree, WIT Finalist (2018, 2019, 2020, 2021, 2022). Dr. Sneha has published over 100 peer-reviewed research; authored a book on revolutionizing healthcare; delivered panel sessions, workshops, and keynotes; in addition to serving on several boards. Dr. Sneha serves as editorial steward and university ambassador for Blockchain in Healthcare Today; editorial board for Health Systems Journal; co-chairs Health IT at AMCIS, and HICSS. Her leadership has led to enhancing KSU’s footprint on diversity and equity, STEAM education, and outreach. She has a PhD in Computer Information Systems from Georgia State University and a BS in Computer Science from University of Maryland. Prior to academia, she worked at PricewaterhouseCoopers in Management Consulting.